Understanding the intricacies of biological systems often involves delving into specialized terminology and concepts. One such concept is the buffer definition biology, which plays a crucial role in maintaining the stability of biological environments. Buffers are essential for regulating pH levels, ensuring that biological processes can occur efficiently and effectively. This post will explore the significance of buffers in biology, their mechanisms, and their applications in various biological contexts.
What is a Buffer in Biology?
A buffer in biology is a solution that resists changes in pH when small amounts of acid or base are added. This resistance is crucial for maintaining the optimal pH range required for biological processes. Buffers achieve this by containing a weak acid and its conjugate base or a weak base and its conjugate acid. These components work together to neutralize any added acids or bases, thereby stabilizing the pH.
Mechanism of Buffers
The mechanism of a buffer involves the equilibrium between a weak acid (HA) and its conjugate base (A-). When an acid is added to the buffer solution, the conjugate base reacts with the added hydrogen ions (H+), neutralizing them and preventing a significant drop in pH. Conversely, when a base is added, the weak acid donates hydrogen ions to neutralize the hydroxide ions (OH-), preventing a significant rise in pH.
The equilibrium can be represented as:
HA ⇌ H+ + A-
When an acid is added:
A- + H+ → HA
When a base is added:
HA → H+ + A-
OH- + H+ → H2O
Importance of Buffers in Biological Systems
Buffers are vital in various biological systems for several reasons:
- Maintaining pH Homeostasis: Biological processes are highly sensitive to pH changes. Buffers help maintain the optimal pH range, ensuring that enzymes and other biological molecules function correctly.
- Protecting Cells and Tissues: Buffers protect cells and tissues from the damaging effects of pH fluctuations, which can disrupt cellular functions and lead to cell death.
- Enhancing Enzyme Activity: Many enzymes require a specific pH range to function optimally. Buffers ensure that this range is maintained, enhancing enzyme activity and efficiency.
- Supporting Metabolic Processes: Metabolic processes, such as glycolysis and the Krebs cycle, rely on specific pH conditions. Buffers help maintain these conditions, supporting overall metabolic health.
Types of Buffers in Biology
There are several types of buffers used in biological systems, each with its unique properties and applications. Some of the most common types include:
- Phosphate Buffers: These buffers are commonly used in biological research and are effective in the pH range of 5.8 to 8.0. They are particularly useful in molecular biology and biochemistry experiments.
- Tris Buffers: Tris (tris(hydroxymethyl)aminomethane) buffers are widely used in molecular biology for their ability to maintain a stable pH around 7.0 to 9.0. They are commonly used in DNA and protein purification processes.
- HEPES Buffers: HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid) buffers are effective in the pH range of 6.8 to 8.2 and are often used in cell culture and physiological studies.
- Carbonate Buffers: These buffers are important in maintaining the pH of blood and other bodily fluids. They play a crucial role in the body's acid-base balance.
Applications of Buffers in Biology
Buffers have a wide range of applications in biology, from research and diagnostics to therapeutic interventions. Some key applications include:
- Cell Culture: Buffers are essential in cell culture media to maintain the optimal pH for cell growth and proliferation. Common buffers used in cell culture include HEPES and bicarbonate.
- Molecular Biology: Buffers are used in various molecular biology techniques, such as PCR, gel electrophoresis, and protein purification. They help maintain the pH required for these processes to be effective.
- Diagnostics: Buffers are used in diagnostic tests to ensure accurate results. For example, buffers are used in blood tests to maintain the pH of the sample, preventing false results.
- Therapeutic Interventions: Buffers are used in therapeutic interventions to maintain the pH of bodily fluids and tissues. For example, buffers are used in dialysis solutions to maintain the pH of the blood during dialysis.
Buffer Definition Biology in Human Physiology
In human physiology, buffers play a critical role in maintaining the body's acid-base balance. The body's buffering systems include:
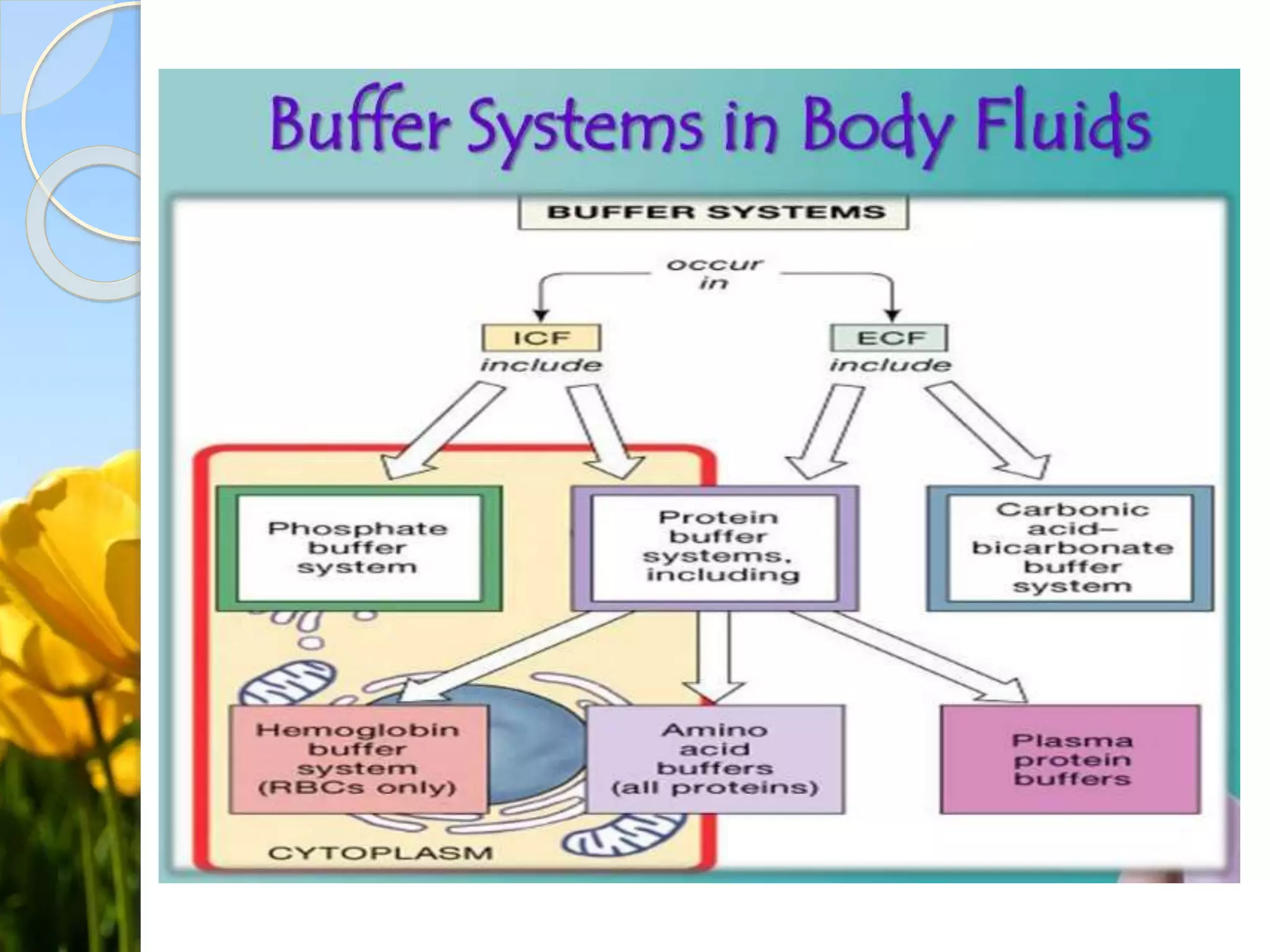
- Bicarbonate Buffer System: This is the primary buffering system in the body, responsible for maintaining the pH of blood and other bodily fluids. It involves the equilibrium between carbonic acid (H2CO3) and bicarbonate (HCO3-).
- Phosphate Buffer System: This system is important in maintaining the pH of intracellular fluids and urine. It involves the equilibrium between phosphoric acid (H3PO4) and its conjugate bases.
- Protein Buffer System: Proteins, particularly hemoglobin in red blood cells, act as buffers by binding hydrogen ions and preventing significant pH changes.
These buffering systems work together to maintain the body's pH within a narrow range, typically between 7.35 and 7.45. Any deviation from this range can lead to serious health issues, such as acidosis or alkalosis.
Buffer Definition Biology in Environmental Science
Buffers are not only important in biological systems but also play a crucial role in environmental science. They help maintain the pH of natural waters, such as lakes, rivers, and oceans, which is essential for the survival of aquatic organisms. For example, the carbonate buffer system in seawater helps maintain the pH, which is critical for the survival of marine life, including corals and shellfish.
In soil science, buffers help maintain the pH of soil, which is important for plant growth and nutrient availability. Soil buffers, such as organic matter and clay minerals, can neutralize acids and bases, preventing significant pH changes that could harm plants.
Buffer Definition Biology in Industrial Applications
Buffers have numerous industrial applications, particularly in the food, pharmaceutical, and chemical industries. They are used to maintain the pH of products, ensuring their stability and efficacy. For example, buffers are used in the production of beverages to maintain their pH, preventing spoilage and ensuring consistent taste. In the pharmaceutical industry, buffers are used in the formulation of drugs to maintain their pH, ensuring their stability and effectiveness.
In the chemical industry, buffers are used in various processes, such as catalysis and synthesis, to maintain the pH required for these processes to be effective. They help ensure that chemical reactions occur under optimal conditions, enhancing efficiency and yield.
Buffer Definition Biology in Research and Development
Buffers are essential tools in research and development, particularly in the fields of biochemistry, molecular biology, and physiology. They are used in various experimental techniques to maintain the pH required for these techniques to be effective. For example, buffers are used in enzyme assays to maintain the pH required for enzyme activity, ensuring accurate results. In molecular biology, buffers are used in techniques such as PCR and gel electrophoresis to maintain the pH required for these techniques to be effective.
In physiological research, buffers are used to maintain the pH of experimental solutions, ensuring that physiological processes can be studied under optimal conditions. This is particularly important in studies involving cell culture, tissue culture, and organ culture, where maintaining the pH is crucial for the survival and function of cells and tissues.
Buffer Definition Biology in Education
Understanding the buffer definition biology is fundamental in educational settings, particularly in biology, chemistry, and environmental science courses. Students learn about the importance of buffers in maintaining pH homeostasis, their mechanisms, and their applications in various fields. This knowledge is essential for students pursuing careers in science, medicine, and environmental science.
In educational settings, buffers are often used in laboratory experiments to demonstrate their role in maintaining pH. For example, students may conduct experiments to observe the effects of adding acids and bases to buffer solutions and measure the resulting pH changes. These experiments help students understand the principles of buffering and their importance in biological systems.
In addition to laboratory experiments, buffers are often discussed in lectures and textbooks, providing students with a theoretical understanding of their role in biology. This knowledge is essential for students to comprehend the complexities of biological systems and their regulation.
In summary, buffers are essential components of biological systems, playing a crucial role in maintaining pH homeostasis. They are used in various applications, from research and diagnostics to therapeutic interventions and industrial processes. Understanding the buffer definition biology is fundamental for students and professionals in the fields of biology, chemistry, and environmental science.
📝 Note: The information provided in this post is for educational purposes only and should not be used as a substitute for professional advice. Always consult with a qualified professional for specific questions or concerns related to biology and buffering systems.
In conclusion, buffers are indispensable in biological systems, ensuring that pH levels remain within optimal ranges for various processes to occur efficiently. From maintaining cellular functions to supporting metabolic processes, buffers play a pivotal role in the stability and health of biological environments. Their applications extend beyond biology into environmental science, industrial processes, and educational settings, highlighting their versatility and importance. Understanding the buffer definition biology provides a foundational knowledge that is essential for advancing in various scientific and medical fields.
Related Terms:
- how does a buffer work
- why are buffers biologically important
- role of buffers
- what are buffers in anatomy
- biological buffers examples
- what are buffers used for