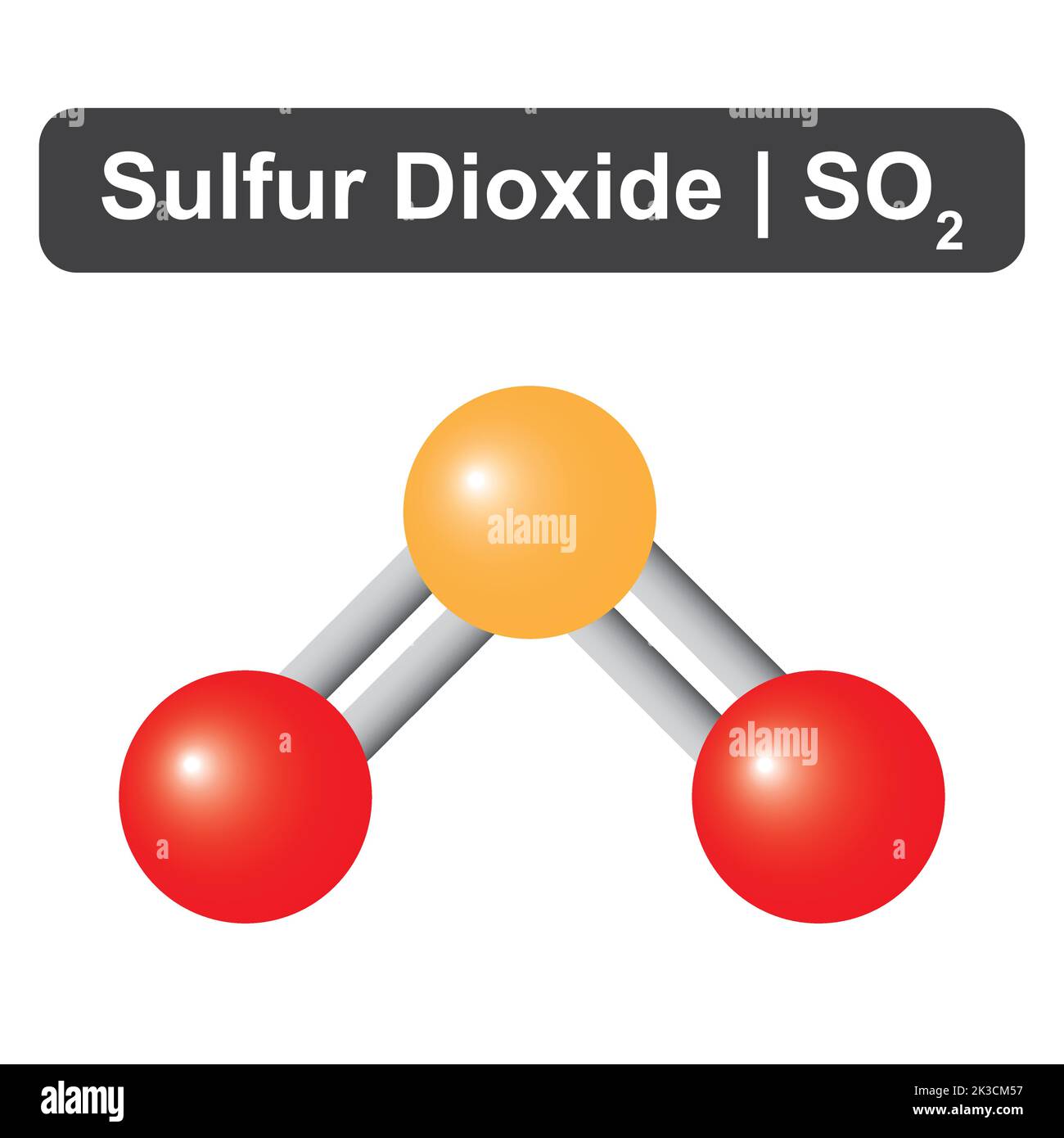
Sulfur dioxide (SO2) is a crucial compound in both industrial and environmental contexts. Understanding its molecular structure, particularly the bond angle of SO2, is essential for comprehending its chemical behavior and reactivity. This blog post delves into the intricacies of the SO2 molecule, exploring its geometry, bond angles, and the factors influencing these properties.
Understanding the Molecular Structure of SO2
Sulfur dioxide is a linear molecule consisting of one sulfur atom bonded to two oxygen atoms. The sulfur atom is the central atom, and the oxygen atoms are bonded to it through double bonds. The molecular formula for sulfur dioxide is SO2, and it has a bent shape due to the presence of lone pairs on the sulfur atom.
The Bond Angle of SO2
The bond angle of SO2 is approximately 119.5 degrees. This angle is a result of the molecule's electronic structure and the presence of lone pairs on the sulfur atom. The bond angle is crucial for understanding the molecule's reactivity and its interactions with other compounds.
To understand why the bond angle of SO2 is 119.5 degrees, we need to delve into the Valence Shell Electron Pair Repulsion (VSEPR) theory. According to VSEPR theory, the geometry of a molecule is determined by the repulsion between electron pairs in the valence shell of the central atom. In the case of SO2, the sulfur atom has two bonding pairs and one lone pair of electrons.
The presence of the lone pair causes the bonding pairs to be pushed closer together, resulting in a bond angle that is less than the ideal 120 degrees for a trigonal planar geometry. This repulsion leads to the observed bond angle of approximately 119.5 degrees.
Factors Influencing the Bond Angle of SO2
Several factors influence the bond angle of SO2. These include:
- Electronegativity of Oxygen: Oxygen is more electronegative than sulfur, which means it pulls the shared electrons more strongly towards itself. This unequal distribution of electrons affects the bond angle.
- Lone Pairs on Sulfur: The lone pair of electrons on the sulfur atom repels the bonding pairs, causing the bond angle to deviate from the ideal 120 degrees.
- Hybridization of Sulfur: The sulfur atom in SO2 is sp2 hybridized, which means it has three sp2 hybrid orbitals. The lone pair occupies one of these orbitals, and the two bonding pairs occupy the other two. This hybridization contributes to the bent shape and the specific bond angle.
Comparative Analysis with Other Molecules
To better understand the bond angle of SO2, it is helpful to compare it with other molecules that have similar structures. For example, carbon dioxide (CO2) has a linear structure with a bond angle of 180 degrees. This is because carbon dioxide has no lone pairs on the central carbon atom, allowing the bonding pairs to be as far apart as possible.
Water (H2O) is another molecule with a bent shape, similar to SO2. The bond angle in water is approximately 104.5 degrees. This is due to the presence of two lone pairs on the oxygen atom, which repel the bonding pairs more strongly than in SO2, resulting in a smaller bond angle.
Here is a comparative table of bond angles for SO2, CO2, and H2O:
| Molecule | Bond Angle | Central Atom Hybridization | Lone Pairs on Central Atom |
|---|---|---|---|
| SO2 | 119.5 degrees | sp2 | 1 |
| CO2 | 180 degrees | sp | 0 |
| H2O | 104.5 degrees | sp3 | 2 |
Applications and Importance of SO2
Sulfur dioxide has numerous applications in various industries. It is commonly used in the production of sulfuric acid, which is a key component in many chemical processes. SO2 is also used as a preservative in the food industry and as a bleaching agent in the paper industry.
However, SO2 is also a significant environmental pollutant. It is a major contributor to acid rain, which can have detrimental effects on ecosystems and infrastructure. Understanding the bond angle of SO2 and its molecular structure is crucial for developing strategies to mitigate its environmental impact.
In the context of environmental science, the bond angle of SO2 plays a role in its reactivity and how it interacts with other compounds in the atmosphere. For example, SO2 can react with water and oxygen to form sulfuric acid, which is a key component of acid rain. The bond angle influences the molecule's ability to undergo these reactions, affecting its environmental impact.
In industrial settings, the bond angle of SO2 is important for optimizing chemical processes. For instance, in the production of sulfuric acid, understanding the molecular structure of SO2 can help in designing more efficient catalysts and reaction conditions.
📝 Note: The bond angle of SO2 is a critical parameter in both environmental and industrial contexts, influencing its reactivity and interactions with other compounds.
Experimental Determination of the Bond Angle of SO2
The bond angle of SO2 can be determined experimentally using various techniques. One common method is X-ray crystallography, which provides detailed information about the molecular structure, including bond angles and lengths. Another technique is infrared spectroscopy, which can be used to study the vibrational modes of the molecule and infer its geometry.
Infrared spectroscopy is particularly useful for studying the bond angle of SO2 because it can provide information about the bending vibrations of the molecule. The frequency of these vibrations is sensitive to the bond angle, allowing researchers to determine it with high precision.
Experimental techniques are essential for validating theoretical predictions and providing accurate data for further research. By combining experimental and theoretical approaches, scientists can gain a comprehensive understanding of the molecular structure and properties of SO2.
📝 Note: Experimental techniques such as X-ray crystallography and infrared spectroscopy are crucial for determining the bond angle of SO2 and validating theoretical predictions.
In conclusion, the bond angle of SO2 is a fundamental property that influences its chemical behavior and reactivity. Understanding this angle, along with the factors that influence it, is essential for both industrial applications and environmental studies. By exploring the molecular structure of SO2 and comparing it with other molecules, we can gain insights into its unique properties and develop strategies to mitigate its environmental impact. The bond angle of SO2 is a critical parameter that plays a significant role in its interactions with other compounds and its overall reactivity.
Related Terms:
- bond angle of so3
- bond angle of h2o
- bond angle of sf4
- bond angle of bf3
- so2 lewis structure bond angle
- bond angle of of2